The medical field is one industry that requires the utmost precision and accuracy, and the same applies to medical device translation.
Translators need a strong medical background and must stay up to date with terminology and regulations to meet quality and compliance standards.
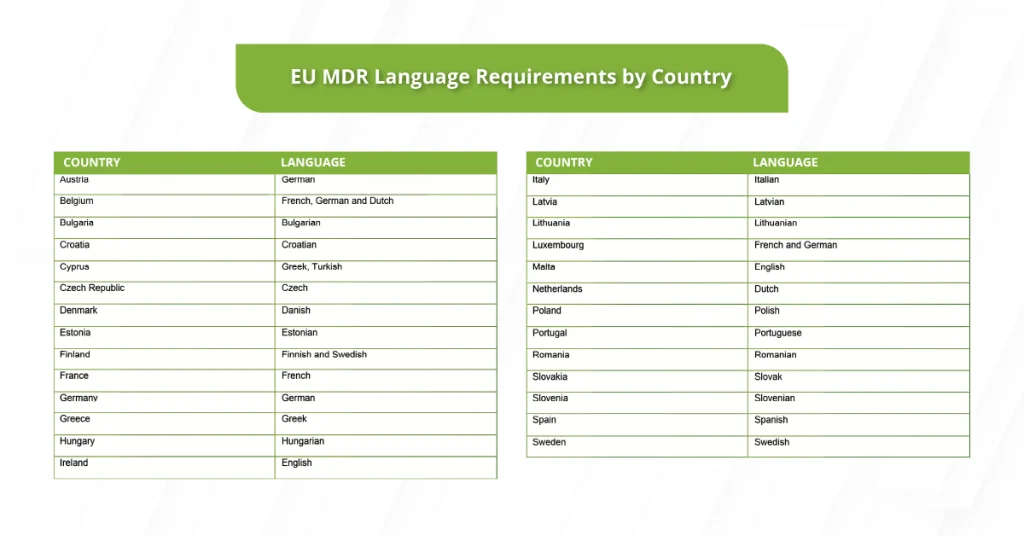
When it comes to medical device regulations, requirements also vary from one country to another.
If your medical services are expanding into the European market, you should follow each Member State’s checklist of required official languages.
Medical device documentation translated into the wrong language can create compliance risks and cause costly delays.
In this guide, you’ll learn the EU MDR (Medical Device Regulation) translation requirements and the key differences across Member States.
What You Need to Know about EU MDR Translation Requirements
As a medical device provider, it’s important to know that the EU Medical Device Regulation (MDR) expects device information to be available in the official EU language(s) required by each Member State.
That means if you supply medical devices in one to three countries, you should translate the relevant documentation and content for those specific markets.
And if you sell EU-wide, you’ll need a language plan that covers the Member State requirements across the countries where your devices are made available.
A common myth is that you must translate everything into every EU language. That’s not true.
Language requirements for medical device documentation vary by Member State and can also depend on the document type.
Why Missing Medical Device Documents Can Block Market Access
So, what medical device documents typically need translation? Here’s a list:
- Labeling and packaging text
- Instructions for Use (IFU)
- Declarations of Conformity
- Technical documentation (often required for audits or requests, depending on the route to market and local expectations)
- Summary of Safety and Clinical Performance (SSCP) (when applicable)
- Field Safety Notices (post-market safety communications)
A single missing language version can lead to product holds, corrective actions, or loss of trust with healthcare professionals. Your device can be safe, but if users can’t understand the safety information, regulators may treat it as unsafe.
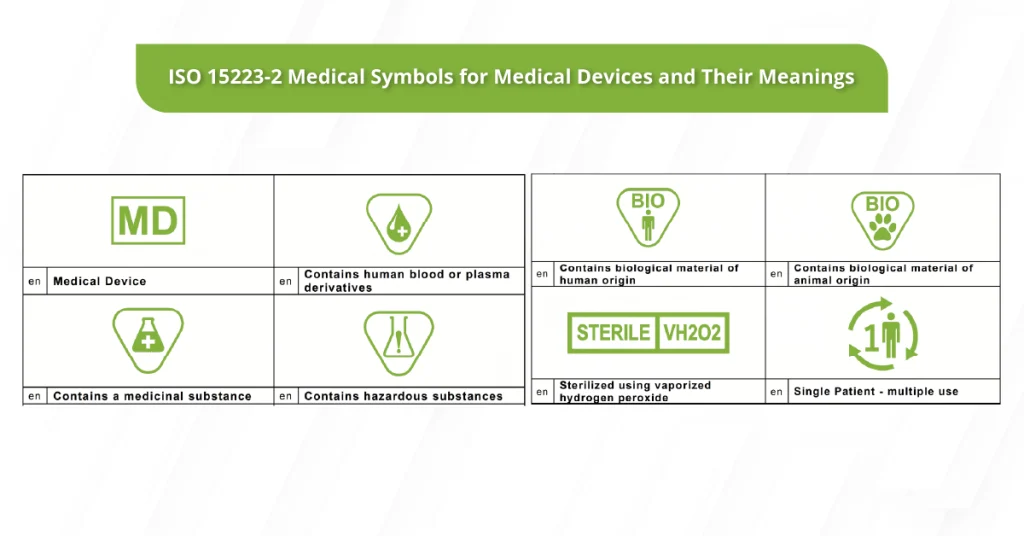
Don’t Forget the Device Interface: GUI Language Can Become a Compliance Issue
GUI (graphic user interface) includes the screens, menus, prompts, and warnings inside a device’s software.
The language used in the GUI—especially warnings and safety instructions—should align with what the user receives to support safe use.
While some countries may accept English for professional use in certain contexts, it’s still best practice to provide the GUI content in the language(s) required by each European country where the device is marketed.
For example, if the IFU is translated but an in-device warning remains in English, you’ve created a usability and safety gap.
Low-Quality Translation Can Become a Safety and Liability Problem
Given the sensitive nature of medical device documentation and on-device text, translation becomes a safety measure not just language support.
Accurate translations protect users from misreading warnings, prevent misuse, and support safe operation in real conditions.
And when something goes wrong, the language version becomes part of what regulators and legal teams look at. If the translation is unclear, inconsistent, or outdated, it can quickly turn into a liability issue.
Under EU MDR expectations, translation should sit inside a quality management system and the broader management systems that control how content is created and maintained.
That includes document control, versioning, approval sign-offs, and traceability of changes across all language versions, so updates in the source don’t create mismatches in the translated files.
This matters even more after the shift from the Medical Device Directive (MDD) to the MDR. MDR comes with more documentation and higher scrutiny, which means language errors are more visible and less forgivable.
A Simple Workflow to Manage EU MDR Translations Across Multiple Languages
Use this checklist to keep your medical device translation processes predictable and audit-ready:
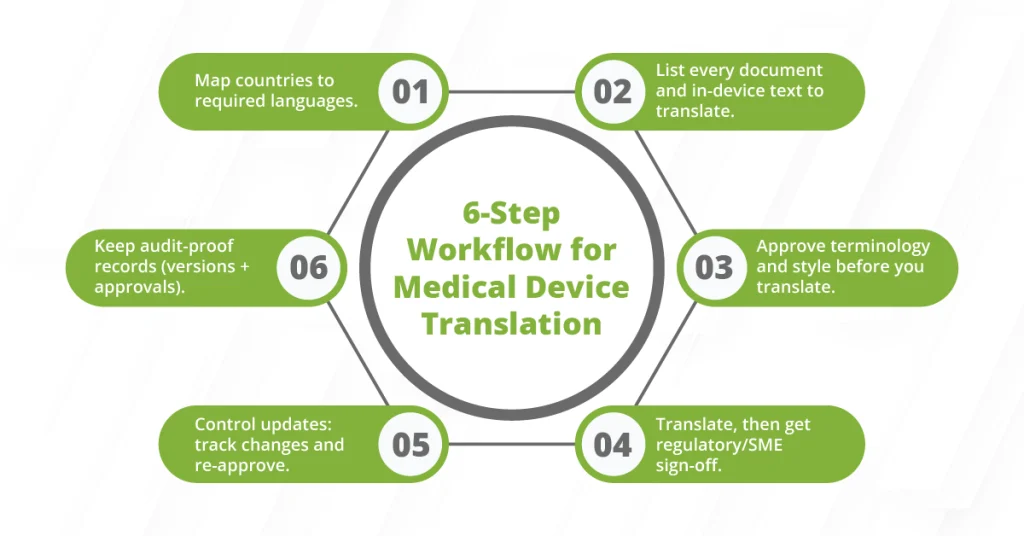
1) Build a country-language matrix:
List where you sell, then map each market to the official languages required for medical device information. This becomes your source of what gets translated and what doesn’t.
2) Inventory what you need to translate:
Separate regulated content from everything else. Include labeling, IFU, SSCP (when applicable), DoC, and any safety text inside software. Then, add the web pages and support content you need to manage content consistently across markets.
3) Lock terminology before translation starts:
Create a glossary and style rules for medical terms, warnings, and units. This is the foundation for managing localized assets across teams without constant disputes.
4) Run a controlled localization process with documented review:
Use defined translation processes: a professional linguist first, then a regulatory or SME reviewer for sign-off. Don’t rely on informal feedback in email threads.
5) Maintain updates through change control:
When the source changes, track what changed, update only the impacted segments, and publish only approved localized versions. No exceptions for “small edits.”
6) Store evidence for audits:
Keep records of who approved what, when, and which version was released. If you can’t prove control, regulators assume you don’t have it.
How Translation Memory and CAT Tools Help You Stay Consistent Across Updates
Translation technology, like translation memory and CAT tools, makes the translation process much easier and more optimized.
Translation memory reuses approved similar translations, and CAT tools have built-in glossaries to enforce consistent wording.
Machine translation is a great asset as well; however, its usage comes with conditions to ensure optimum quality and accuracy.
For example, it’s best to use machine translation as a first draft for low-risk content. Then, this content should be revised by professional translators to validate accuracy and fix any mistranslations.
By using translation tools, you get measurable outcomes, such as fewer inconsistencies and faster updates.
Real World Examples: What Changes When You Expand From One EU Country to Five
In the real world, EU expansion doesn’t add “a few more translations.” It changes how you plan, review, and release.
Scenario A: Launching in 1 country
Fewer official languages apply, approvals are simpler, and it’s easier to keep IFU, labeling, and GUI aligned.
Scenario B: Adding 3–5 member states
Complexity multiplies. Member states may accept different languages, and expectations often differ for patient-facing information versus content intended for healthcare professionals.
Scenario C: Pan-EU growth
At this stage, localization project management becomes mandatory, not optional.
So what changes in practice?
Before shipping, all regulated documents must be finalized in the required languages and any safety-critical interface text.
Patient-facing content typically requires full localization, while some member states may accept limited English for healthcare professionals, but this must be verified country by country.
To avoid mismatches between IFU and GUI, both must follow the same approved source, terminology, and change-control process across all localized versions.
Need MDR-Ready Translations Without Rework?
If you’re preparing IFUs and labeling, our Medical Translation Services team can help you map Member State language requirements and deliver audit-ready files with documented review.
For patient-facing instructions, bayantech provides IFU Translation so your safety content is clear in every target market.
And if you’re scaling across regulated markets, order our Life Sciences Translation services for consistent terminology and controlled workflows across every release.







